Draw a Labelled Diagram of a Nephron
Affiliate v Renal physiology
5.i Torso fluids and their distribution
Water accounts for lx% of body mass in males, i.e., over 40 L in a 70 kg adult. This fraction is somewhat lower in females, at about l%, because of the increased boilerplate trunk content of fat tissue, which contains less water than other tissue types. Full body water is divided between intracellular (two-thirds of total torso fluid) and extracellular compartments (ane-tertiary of total body fluid), with plasma forming a major subcompartment (one-quarter of extracellular fluid) inside the extracellular space (Section i.ane, Fig. 2). Water can motion freely betwixt these fluid spaces and this exchange is controlled by pressure gradients across the jail cell membrane (which separates intracellular and extracellular spaces) and the capillary wall (which separates plasma from interstitial fluid). In each instance, the distribution of fluid depends on the cyberspace effect of hydrostatic and osmotic forces. The solutes controlling osmosis at the ii sites, yet, are quite different. Only those species which cannot diffuse from one compartment to another will exert an osmotic result (Section 1.3) and the relevant barriers to diffusion demonstrate very dissimilar solute permeabilities.
Ionic limerick of extracellular fluid
The extracellular concentrations of a number of important ions are commonly adamant using plasma from venous blood samples. Normal values are summarized in Table 8. Although the total charge carried by all the cations and anions must be equal, a significant fraction of the anion load is not accounted for in routine measurements. Information technology can exist seen that the sum of the measured cations exceeds the sum of the measured anions, i.e.,
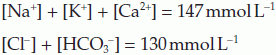
The difference between these totals is called the anion gap (17 mmol L−one in this case). This represents the contribution made past anions non normally measured in the clinical laboratory, eastward.g., sulphate, phosphate, proteins and other organic anions. Any condition which generates additional anions of this sort, e.g., the anions released by dissociation of ketoacids in diabetes mellitus (Section 8.seven), will increase the anion gap.
5.2 Relevant renal structure
There are two kidneys, each with an outer cortex and inner medulla (Fig. 78A). Urine is formed within functional subunits known as nephrons (Fig. 78B). Each nephron contains a glomerulus, consisting of a tuft of capillaries, along with the afferent and efferent arterioles through which blood enters and leaves (Fig. 78C). The glomerulus is surrounded past the epithelium of the Bowman's capsule and these structures combine to form the renal corpuscle. This is the site of initial plasma filtration. The resulting filtrate is then modified by a variety of secretory and reabsorptive processes as it passes in plough through the proximal convoluted tubule, the loop of Henle, the distal convoluted tubule and the collecting duct (Fig. 78B). The glomeruli and convoluted tubules lie within the renal cortex while the loops of Henle and collecting ducts extend down into the medullary region. The finish product, urine, is eventually delivered via the renal pelvis to the ureter, which empties into the bladder.
Ultrastructural features of the nephron
The walls of each nephron are fabricated up of a unmarried layer of epithelial cells, although the appearance of these cells is not compatible throughout. Specialized epithelial cells in Bowman'south sheathing called podocytes surround the glomerular capillaries and contribute to the filtration bulwark (Department 5.3, Fig. 79). The cells lining the convoluted tubules acquit a brush border of microvilli on their luminal surface and contain big numbers of intracellular mitochondria. These features are peculiarly pronounced in the proximal convoluted tubule and provide a large surface area and rich supplies of ATP, appropriate features for a region specializing in active transport. In the descending limb of the loop of Henle, by dissimilarity, the epithelium is flat, with few organelles. This is consistent with a depression level of active transport and high permeability to fluid and electrolytes. Cells in the more than distal, thick portion of the ascending limb, however, are more similar those in the tubules. These cells are involved in agile ion transport and evidence a very depression level of permeability to water, probably because of well-developed tight junctions between adjacent epithelial cells in this surface area.
five.4 Modification of glomerular filtrate
The formation of urine can exist thought of as a ii-phase procedure. Filtration produces an essentially protein-free, but otherwise plasma-like fluid with an osmolality of virtually 300 mosmol kg−one. This is and then modified by reabsorption and secretion as it passes through the balance of the nephron. Some of these processes are hormonally controlled and this allows the volume, ionic composition and total osmolality of urine to be varied in accordance with the homeostatic needs of the body.
Mechanisms leading to reabsorption of Na + and water
Each of the main structural components of the nephron contributes to control of the extracellular fluid volume and composition through mechanisms which pb to the reabsorption of Na+ and water.
Distal convoluted tubule and collecting ducts
There are 2 important mechanisms which play a role in Na+ and water reabsorption in these regions.
The countercurrent multiplier mechanism in the loop of Henle
Ion transport in the loop of Henle is responsible for the generation of the very hypertonic weather inside the renal medulla, with osmolalities reaching a maximum of virtually 1200 mosmol kg−1. Although the principles underlying this have already been discussed, i.e., active reabsorption of Na+ in the thick ascending limb, with little or no parallel movement of water, and free improvidence across the walls of the descending limb, we will at present briefly consider the countercurrent multiplier machinery, which helps explain the very high osmolalities accomplished. This reflects a blueprint which effectively allows filtered fluid to be concentrated not one time merely several times as information technology moves round the loop.
Let the states presume that the system is initially filled with isotonic solution (300 mosmol kg−i) from the proximal convoluted tubules. The ion pumps will then transfer solute from the ascending limb into the interstitium until the concentration gradient generated across the epithelium exactly opposes the energy available from the active transport system (Section 1.3). In a simple, single conduit system, this might limit the slope to about 200 mosmol kg−one, with a maximal interstitial osmolality of nigh 400 mosmol kg−1 (Fig. 83A). In the countercurrent system, however, the solution in the permeable descending limb equilibrates with the interstitium then becomes concentrated. As more (isotonic) fluid enters at the top of the descending limb, concentrated fluid is forced round into the ascending limb, reducing the transepithelial osmotic gradient while maintaining a loftier total interstitial osmolality (Fig. 83B). Ion pumping tin proceed once more, until a 200 mosmol kg−i gradient is again achieved. In this example, the osmolality in the medullary interstitium and lower part of the descending limb is raised to 500 mosmol kg−1 equally a upshot. Repeated cycles of ion pumping followed by fluid transfer can thus generate a higher medullary osmolality than would otherwise be possible. At the aforementioned time, a slope in osmolality develops along the length of the loop of Henle with low values in the upper medulla and loftier values next to its tip.
Ship of H + and HCO3 −
The kidney plays an important role in the regulation of extracellular pH through several dissimilar, if related, tubular mechanisms :
These processes lead to the excretion of backlog H+ or HCO3 − in the urine and the resulting urinary pH can vary between iv.5 and 8.2, depending on the conditions. Under normal circumstances the terminate upshot is reabsorption of all filtered HCOthree − within the proximal convoluted tubule, and urinary excretion of an acid load equivalent to 50–80 mmol of H+ each twenty-four hour period, by and large every bit HtwoPO4 − and NHfour + formed within the distal convoluted tubule. This metabolic acid is generated during catabolism of the proteins in an average mixed nutrition. Cyberspace excretion of acid is coupled with tubular generation of boosted HCOiii −, thus tending to enhance plasma [HCO3 −].
H+ secretion and HCOthree − absorption
Bicarbonate is one of the well-nigh important pH buffers in the body (Section five.ix). Since it is freely filtered in the glomerulus it is vital that the renal tubules should take an efficient mechanism for HCOiii − absorption, otherwise its loss in the urine would quickly atomic number 82 to fatal acidosis. Protons and HCO3 − are generated within the tubular epithelial cells by dissociation of carbonic acid formed past the reaction of COii with h2o (Fig. 86A). This reaction is catalyzed by the enzyme carbonic anhydrase. The H+ is actively transported into the tubular lumen while the HCO3 − diffuses into the peritubular capillaries, maintaining electrical neutrality within the cell. Secreted H+ has to be buffered in the urine to limit the [H+] gradient between the tubular cells and the lumen, otherwise it would quickly go too large for the active transport system, preventing further secretion. Filtered HCOthree − reacts with H+ to course CO2 and H2O in the tubular fluid. Carbon dioxide can diffuse freely back across the tubular cell membrane, completing the cycle. The cyberspace event is that one HCO3 − ion is captivated into the peritubular plasma from the filtrate for each H+ which is secreted.
This mechanism conserves HCOthree − and, under normal conditions, all the filtered HCOthree − is absorbed (mainly in the proximal convoluted tubule) and none is excreted in urine. If the rate at which HCOiii − is filtered exceeds the rate of H+ secretion, notwithstanding, then the backlog cannot be captivated and will be excreted, making the urine element of group i. This mechanism tends to reduce plasma HCO3 − during alkalosis, and thus tends to lower the pH (meet Eq. 41 in Section 5.9).
H+ excretion and HCOthree − generation
Although the mechanisms described to a higher place couple H+ secretion to HCO3 − absorption, they exercise not atomic number 82 to any internet excretion of H+ in the urine since the CO2 produced diffuses back out of the tubular lumen (Fig. 86A). This underlines the point that renal secretion (a tubular cellular mechanism) and excretion (permanent removal in the urine) are non the same thing. Renal excretion of acrid still depends on tubular secretion but in this instance the H+ is buffered by phosphate species such as HPO4 2−, or ammonia (NH3). The H2PO4 − and ammonium (NH4 +) ions formed cannot cross the lipid membrane of the tubular epithelium and are excreted in the urine (Fig. 86B, C). Phosphate enters the lumen by filtration but the NH3 is actually secreted by the tubular cells, much of it having been synthesized from glutamine inside the cell (ammoniagenesis).
Ane important feature of H+ buffering past phosphate and ammonia is that information technology leaves the HCO3 − produced by dissociation of carbonic acid within the tubular cells to diffuse into the extracellular fluid. Since this amounts to generation of additional HCO3 − over and above that which was filtered in the glomerulus, it can actually raise plasma [HCO3 −], rather than simply preventing it from decreasing as absorption of filtered HCOthree − does. Bicarbonate is an of import base inside the body, and this mechanism provides for renal compensation of a respiratory acidosis. For instance, during a chronic respiratory acidosis both the rate of H+ secretion and the charge per unit of NHthree product are upregulated inside the first week, leading to an increase in plasma [HCOiii −]. This tends to opposite the fall in blood pH, at to the lowest degree partially compensating for the original defect (Sections 4.4 and five.9).
Reabsorption of glucose and other nutrients
Glucose is freely filtered in the glomerulus so the concentration in the filtrate equals that in plasma. This means that the glucose load presented to the tubules is proportional to the plasma glucose concentration (Fig. 87). Normally, the charge per unit of active glucose reabsorption in the proximal convoluted tubule equals the rate of filtration, then no glucose appears in the urine. This reuptake can eventually be saturated, however, and the maximum achievable rate of glucose reabsorption is known as the transport maximum (T max). If the rate of glucose filtration rises to a higher place T max, the backlog load volition all be excreted in the urine (Fig. 87). This occurs when the plasma glucose concentration rises higher up most 11 mmol L−1, so this is regarded as the renal threshold concentration, above which glucose will be excreted in the urine (glycosuria). Glycosuria is an important feature of diabetes mellitus, in which a deficiency of the hormone insulin leads to abnormally high glucose concentrations (Section 8.seven).
The relationship betwixt plasma glucose concentration and urinary excretion rate is curved over a range of concentrations immediately above threshold. This region of splay reflects small variations in the maximum transport charge per unit for different tubules within the kidneys. Somewhen all glucose transport systems are saturated, and urinary excretion then rises linearly with plasma glucose since any increment in the filtered glucose load now results in an equal increment in glucose excretion.
Tubular maximum send mechanisms of this sort are likewise involved in the reabsorption of other filtered nutrients from the proximal convoluted tubule, especially amino acids. Systems of this kind should exist contrasted with the gradient–time dependent resorption of Na+, in which a stock-still fraction of the filtered load is reabsorbed (Fig. 82).
Source: https://basicmedicalkey.com/renal-physiology/
0 Response to "Draw a Labelled Diagram of a Nephron"
Postar um comentário